Right about now you might be asking yourself, "what's so important about hydrogen?" Its a good question, and the answer is: everything! But, for now, lets start with the basics.
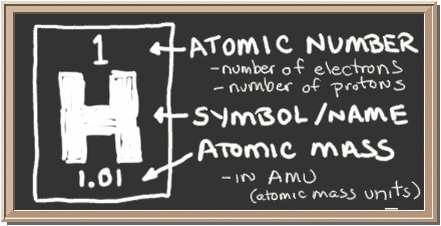
Don't let the periodic table fool you, it isn't difficult to understand. As you can see from this entry, Hydrogen has an Atomic Number of 1. This means that a regular hydrogen atom has 1 proton, and 1 electron. The Atomic Symbol for Hydrogen is "H". The Atomic Mass of Hydrogen is 1.01 AMU. Don't let Atomic Mass Units confuse you. AMUs are a measurement of weight just like ounces or pounds. Atoms are too small to be measured by ounces, however, so we came up with a smaller unit of measurement. As you can probably guess, AMUs we're created based off of the weight of 1 Hydrogen atom, and thus Hydrogen's Atomic Mass is very close to 1.
You might be asking why Hydrogen's Atomic Mass isn't exactly 1. If you're curious, you can click here to find out, or you can scroll down to the section that talks about Isotopes. If you've ever heard of the Hindenburg Disaster, you'll know that Hydrogen is actually lighter than air, and that it floats just like helium. That's because the Atomic Mass of Hydrogen is so small compared to the rest of the atoms on the periodic table. However, AMUs are actually a median measure of all of an elements isotopes, and thus Hydrogen's atomic mass isn't exactly 1.
Don't let the periodic table fool you, it isn't difficult to understand. As you can see from this entry, Hydrogen has an Atomic Number of 1. This means that a regular hydrogen atom has 1 proton, and 1 electron. The Atomic Symbol for Hydrogen is "H". The Atomic Mass of Hydrogen is 1.01 AMU. Don't let Atomic Mass Units confuse you. AMUs are a measurement of weight just like ounces or pounds. Atoms are too small to be measured by ounces, however, so we came up with a smaller unit of measurement. As you can probably guess, AMUs we're created based off of the weight of 1 Hydrogen atom, and thus Hydrogen's Atomic Mass is very close to 1.
You might be asking why Hydrogen's Atomic Mass isn't exactly 1. If you're curious, you can click here to find out, or you can scroll down to the section that talks about Isotopes. If you've ever heard of the Hindenburg Disaster, you'll know that Hydrogen is actually lighter than air, and that it floats just like helium. That's because the Atomic Mass of Hydrogen is so small compared to the rest of the atoms on the periodic table. However, AMUs are actually a median measure of all of an elements isotopes, and thus Hydrogen's atomic mass isn't exactly 1.
Atomic Structure
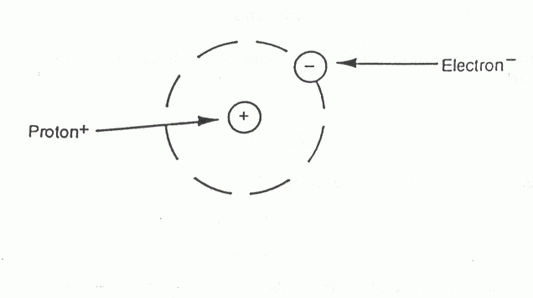
In the simplest sense an atom is a proton with an electron spinning around it. In between these two subatomic (smaller than an atom) particles is a lot of empty space.
There are many subatomic particles, and not all of them bond together to form atoms. For instance, a photon of light is a subatomic particle. But, the three subatomic particles that form atoms are protons, neutrons, and electrons. These particles all have mass, and you may find it easier to think of them as tiny little magnets.The electron is a little negatively charged magnet, and the proton is a little positively charged magnet that holds the electron close.
If negative electrons are attracted to the positive charge of protons, you can guess that electrons are repelled by the negative charge of other electrons. They can't stand each other. When two atoms get too close to each other, the electrons in each atom repel. If that were the end of the story, however, nothing orderly in the Universe would ever form. After all, you're made up of trillions of trillions of atoms and they seem to stick together just fine. The Earth is made up of atoms, and it sticks together. Even the gas in the nebula tends to clump together. So what's the story?
There are many subatomic particles, and not all of them bond together to form atoms. For instance, a photon of light is a subatomic particle. But, the three subatomic particles that form atoms are protons, neutrons, and electrons. These particles all have mass, and you may find it easier to think of them as tiny little magnets.The electron is a little negatively charged magnet, and the proton is a little positively charged magnet that holds the electron close.
If negative electrons are attracted to the positive charge of protons, you can guess that electrons are repelled by the negative charge of other electrons. They can't stand each other. When two atoms get too close to each other, the electrons in each atom repel. If that were the end of the story, however, nothing orderly in the Universe would ever form. After all, you're made up of trillions of trillions of atoms and they seem to stick together just fine. The Earth is made up of atoms, and it sticks together. Even the gas in the nebula tends to clump together. So what's the story?
Atomic Bonds
When most people think of Chemistry, they think of complicated equations, lab explosions, and maybe even fuels or alcohols which have chemical-sounding names like Methane and Ethanol. But Chemistry on the most fundamental level is simply the study of how atoms interact with each other. Just remember that atoms are like tiny magnets with an outer shell of negatively charged electrons, and a center - or nucleus - of protons and neutrons (neutrons have no charge at all and we won't be discussing them much yet). So why don't the negative electrons in atoms simply repel the negative electrons in all other atoms?
Covalent Bonding
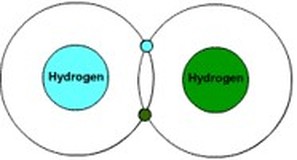
Electons are not particular about which proton they are attracted to. In other words, an electron in one hydrogen atom is attracted to another hydrogen atom's protons, assuming they get close enough to each other. Magnetism, like gravity, weakens rapidly as distance increases. However, if two atoms come into contact with each other, they can sometimes reach a balance, an equilibrium, where the attraction of electrons to both nuclei holds both atoms together. Conveniently, Hydrogen is attracted to Hydrogen in just this way. If one Hydrogen gets close to another, the electrons will actually begin to spin around both nuclei. In other words, two Hydrogen atoms will "share" their electrons with each other. This is called covalent bonding. The chemical that results when two Hydrogen atoms get close together is written out as H2, or "2 hydrogen atoms".
Any single element with the subscript 2 such as "H2" is called a Diatom (two atoms). Many elements found in nature will bond in this way. In fact, most of the oxygen on Earth including what we breathe is a diatom, written out as O2. Sometimes, three of the same kind of atom can bond. An example of this is O3 (3 oxygen atoms) which form covalent bonds. 03 is more commonly referred to as "Ozone". What's interesting is that though there is only a single atom difference between between 02 and 03, we breathe the first to live, and if we breathe too much of the second we'll die.
Any single element with the subscript 2 such as "H2" is called a Diatom (two atoms). Many elements found in nature will bond in this way. In fact, most of the oxygen on Earth including what we breathe is a diatom, written out as O2. Sometimes, three of the same kind of atom can bond. An example of this is O3 (3 oxygen atoms) which form covalent bonds. 03 is more commonly referred to as "Ozone". What's interesting is that though there is only a single atom difference between between 02 and 03, we breathe the first to live, and if we breathe too much of the second we'll die.
Ionic Bonding
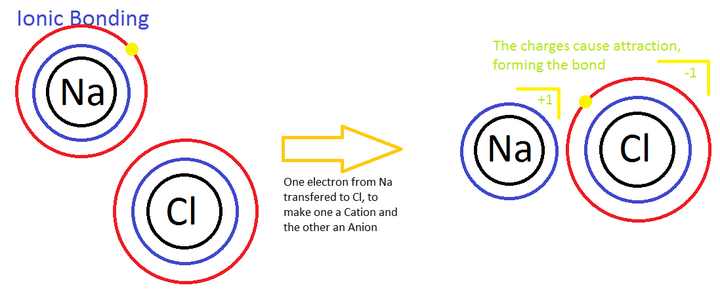
What would happen if a Hydrogen atom lost its one and only electron? Well, the atom would then consist of one proton (+1) and one neutron. The overall charge would then be positive. What would happen if a Hydrogen atom gained an electron? It would consist of its nucleus of 1 proton (+1) and a neutron, but also 2 electrons (-2). The overall charge would then be (-1). When an entire atom has a positive charge, usually from losing an electron, it is called a Cation. When an entire atom has a negative charge, usually from gaining an electron, it is called an Anion. Any atom that has an overall positive or negative charge from gaining or losing electrons is simply called an Ion.
Lets look at another way chemicals can form in the picture on the right. If a sodium atom (Atomic Number 11) and a Chlorine atom (Atomic Number 17) get close to each other, the Chlorine atom will steal an electron from the sodium atom. The Chlorine does not "share" this electron with the sodium, it simply takes it away.
However, because the sodium atom loses one of its electrons, it now has 11 protons (+11) and 10 electrons (-10) left, giving it a +1 charge. Because it has a positive charge we can call it a Cation.
The Chlorine atom gains the electron it steals, and so it has 17 protons (+17) and 18 electrons (-18), giving it a (-1) charge, and we then call it an Anion.
Now, as we learned earlier, opposites attract, and so the entire positively charged sodium atom is now attracted to the entire negatively charged chlorine atom, and this will cause them to bond together. When two atoms with opposite charges bond together, it is called Ionic Bonding. The symbol for this new chemical would be NaCl (1 sodium atom and 1 chlorine atom) and in case you were wondering, that's the chemical formula for table salt.
Lets look at another way chemicals can form in the picture on the right. If a sodium atom (Atomic Number 11) and a Chlorine atom (Atomic Number 17) get close to each other, the Chlorine atom will steal an electron from the sodium atom. The Chlorine does not "share" this electron with the sodium, it simply takes it away.
However, because the sodium atom loses one of its electrons, it now has 11 protons (+11) and 10 electrons (-10) left, giving it a +1 charge. Because it has a positive charge we can call it a Cation.
The Chlorine atom gains the electron it steals, and so it has 17 protons (+17) and 18 electrons (-18), giving it a (-1) charge, and we then call it an Anion.
Now, as we learned earlier, opposites attract, and so the entire positively charged sodium atom is now attracted to the entire negatively charged chlorine atom, and this will cause them to bond together. When two atoms with opposite charges bond together, it is called Ionic Bonding. The symbol for this new chemical would be NaCl (1 sodium atom and 1 chlorine atom) and in case you were wondering, that's the chemical formula for table salt.
An Abundance of Hydrogen.

Now that we've got an idea about what atoms are and how they interact, lets return our focus to one specific type of atom, namely, Hydrogen. To say that Hydrogen is in abundance in the Universe is an understatement. There was a time when the Universe was made almost entirely of Hydrogen, and there is something intuitive about that, isn't there? After all, Hydrogen is 1 on the periodic table, and with 1 proton and 1 electron, it is the simplest atom. Knowing this, it shouldn't be surprising to learn that even now our visible Universe is made up almost entirely of Hydrogen!
Billions of years ago Hydrogen accounted for over 99 percent of all visible matter in the Universe. At that time, the only other elements that existed were Helium (Atomic Number 2) and Lithium (Atomic Number 3) in trace amounts. You might be thinking that on Earth, some of the elements are comparatively rare. After all, you're a Carbon based life form. Was there really a time where the element Carbon (Atomic Number 6) didn't exist? If so, where did Carbon come from? And for that matter, where did the Iron (Atomic Number 26) in our blood, the Oxygen (Atomic Number 8) we breathe, and the Sodium (Atomic Number 11) in the salt shaker come from? Our Universe has changed drastically in the almost 14 billion years since its beginning, but one thing hasn't changed. The visible Universe is still predominantly made up of Hydrogen, about 90 percent. But something happened billions of years ago (just 200-300 Million years after the Big Bang if you are curious), something so drastic that it changed the chemical composition of the entire Universe, and ever since then, the Hydrogen in our Universe has been steadily disappearing! Mind you, this is happening extremely slowly.
Billions of years ago Hydrogen accounted for over 99 percent of all visible matter in the Universe. At that time, the only other elements that existed were Helium (Atomic Number 2) and Lithium (Atomic Number 3) in trace amounts. You might be thinking that on Earth, some of the elements are comparatively rare. After all, you're a Carbon based life form. Was there really a time where the element Carbon (Atomic Number 6) didn't exist? If so, where did Carbon come from? And for that matter, where did the Iron (Atomic Number 26) in our blood, the Oxygen (Atomic Number 8) we breathe, and the Sodium (Atomic Number 11) in the salt shaker come from? Our Universe has changed drastically in the almost 14 billion years since its beginning, but one thing hasn't changed. The visible Universe is still predominantly made up of Hydrogen, about 90 percent. But something happened billions of years ago (just 200-300 Million years after the Big Bang if you are curious), something so drastic that it changed the chemical composition of the entire Universe, and ever since then, the Hydrogen in our Universe has been steadily disappearing! Mind you, this is happening extremely slowly.
The Chemistry of the Universe!
In a sense, the early Universe was a lot like the nebulae we see today, only much hotter and much denser. You can think of the early Universe as a big cloud of super heated gas, mostly hydrogen, that expanded into space like a balloon. However, we already learned a fundamental rule about the Universe, and it was as true then as it is now, namely, stuff clumps together. Just like in the nebulae we see today, the gas in the early universe began clumping together. And then, some 200 million years after the Universe was born, the very first stars ignited, illuminating the dark depths of space.
Before we move on to talk about stars, we need to understand the engine that drives a star, and why Hydrogen is so important. We know that stars are big, and hot, and they produce light, but where does all of that energy come from? The engine which drives every star in the Universe is Nuclear Fusion, and it is through fusion that all of the other elements in the periodic table are made!
Before we move on to talk about stars, we need to understand the engine that drives a star, and why Hydrogen is so important. We know that stars are big, and hot, and they produce light, but where does all of that energy come from? The engine which drives every star in the Universe is Nuclear Fusion, and it is through fusion that all of the other elements in the periodic table are made!
Nuclear Fusion: How do we know we are Starstuff?
Fusion is a physical process that smashes atoms together, forming new atoms and releasing energy. A lot of energy. Right now I would love to tell you that 2 Hydrogen atoms smash together and form 1 helium atom, but unfortunately it isn't quite that simple. Essentially, fusion is a somewhat exotic process because, as we've learned, electrons repel electrons, and protons repel protons. So, it is very difficult to get atoms to get close enough together to fuse. There is actually more than one way to do it, but we're only interested in one, namely Thermonuclear Fusion. This is the kind of fusion that powers stars. We call it that, quite simply, because in order for this type of fusion to work, atoms have to get very, very hot. Thermo (temperature) Nuclear (atoms). Hot atoms.
When you heat up matter, lets say a ball of hydrogen gas, the individual Hydrogen atoms start vibrating and whipping around each other in a frenzy. The hotter the atoms, the more energetic their movement. Now think about the inside of a star where the pressure is enormous and the heat is millions of degrees. The atoms inside the star are getting crushed together by the weight of the star, and the heat is making them want to move chaotically. You can guess that in those conditions, atoms are going to start crashing into each other, and if the right temperature and pressure is present, atoms will hit each other with such force that the electrostatic (the charge of protons and neutrons) repulsion they feel for each other will be overcome, and the atoms will fuse together.
When you heat up matter, lets say a ball of hydrogen gas, the individual Hydrogen atoms start vibrating and whipping around each other in a frenzy. The hotter the atoms, the more energetic their movement. Now think about the inside of a star where the pressure is enormous and the heat is millions of degrees. The atoms inside the star are getting crushed together by the weight of the star, and the heat is making them want to move chaotically. You can guess that in those conditions, atoms are going to start crashing into each other, and if the right temperature and pressure is present, atoms will hit each other with such force that the electrostatic (the charge of protons and neutrons) repulsion they feel for each other will be overcome, and the atoms will fuse together.
Under The Electron Microscope: Fusion explained
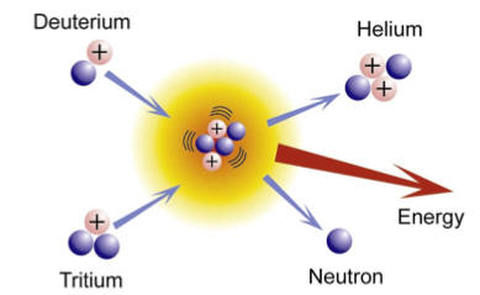
Alright, I know I said we wouldn't talk about neutrons, but its necessary now to explain what you are seeing in this picture. Deuterium and Tritium are isotopes of Hydrogen. The white circles with the plus symbols are Protons, and the purple circles are neutrons. Knowing this, you might be able to figure out what an isotope is. An isotope is an atom of a particular element, in this case Hydrogen, that has a different number of neutrons. Tritium is still Hydrogen, but it has 2 neutrons and one proton. Note that because neutrons are neutral, this additional neutron doesn't change the overall charge of the atom. Deuterium is the name for Hydrogen atoms that have 1 proton and 1 neutron. Before we move on, this isotope stuff may raise more questions than it answers, so lets just look at all possible isotopes of Hydrogen quickly, and then at least we'll have a little background. The good news is, there are only three naturally occurring isotopes of Hydrogen, and we've already talked about two. The three naturally occurring isotopes of Hydrogen are Deuterium, Tritium, and Protium.
Hydrogen Isotopes.
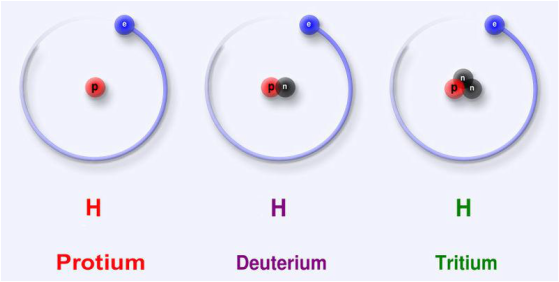
As you can see, all of these isotopes are forms of Hydrogen. The simplest form of Hydrogen is Protium, which consists of just 1 proton and 1 electron. If you add a neutron to a Protium atom, you get a Deuterium atom, and if you add one more neutron, you get a Tritium atom. Again, all of these atoms are still considered hydrogen atoms because they all have the same number of protons and electrons. So now that we know about this little nuance of atoms, lets look at the picture of fusion again.
Thermonuclear Fusion.
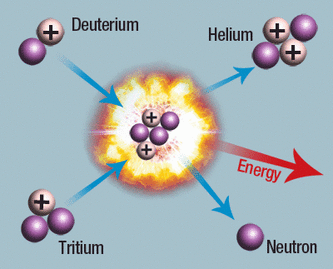
In this model we see one atom of Deuterium (Hydrogen) colliding violently with an atom of Tritium (Hydrogen). After all we've learned about hydrogen and isotopes, what's really important here is the products of this reaction, specifically we see that the result of fusing these two hydrogen atoms is a helium tom and the release of energy. This energy takes the form of electromagnetic radiation (light.) Thus, fusion is the engine which drives stars, producing the heat and light we receive from our Sun. Stars don't stop at fusing hydrogen atoms, they can continue fusing many other atoms like helium and carbon to form even heavier elements. Thus elements like iron and sodium did not exist in the universe until they were formed formed through fusion inside a star. This is how we know we are starstuff, and this is how stars are literally changing the chemistry of the entire Universe! Most of the atoms in your body, in the Earth, and in the Universe started out as Hydrogen, and through fusion many were transformed into the rocky, solid, relatively exotic matter we see today. We'll talk more about stars in the next section, but I'd like to leave you with a little foreboding: Fusion is what gives a star life, and it is also what eventually kills them.
Summary: We are Starstuff.
In summary, Hydrogen was the first element to exist in the Universe. When the Big Bang occurred, conditions were too hot, violent, and chaotic for matter to exist. As the Universe expanded and cooled, tiny subatomic particles started coming together, forming the very first atoms, and since Hydrogen is the simplest atom, it formed in abundance. 200 million years later, the Universe had expanded and cooled enough for huge clouds of hydrogen to collapse, igniting nuclear fusion and forming the first stars. The first stars fused hydrogen into heavier and heavier elements creating the beginnings of the periodic table of elements. Because the Carbon in our bodies didn't even exist until it was formed on the inside of a star, Carl Sagan famously called us "Starstuff".
